The most aggressive type of brain tumor, grade IV glioma known as glioblastoma (GBM), is one of the few tumor types with both a poor outcome and minimal improvement in survival in the past decades. For GBM, like other solid cancers, intratumoral heterogeneity is likely an important factor in mediating therapeutic response. In particular, quiescent, G0-like subpopulations may engender tumors with more robust responses to treatment regimens and allow for tumor regrowth after standard of care (SOC). However, G0-like tumor populations are currently ill-defined, even after application of single cell genomics to GBM. Our failure to fully comprehend and experimentally model quiescent/G0-like states represents a critical knowledge gap, but also a key opportunity, for glioma and other cancers, as neutralizing G0 cells could effectively prevent chemoradiotherapy resistance and tumor recurrence.
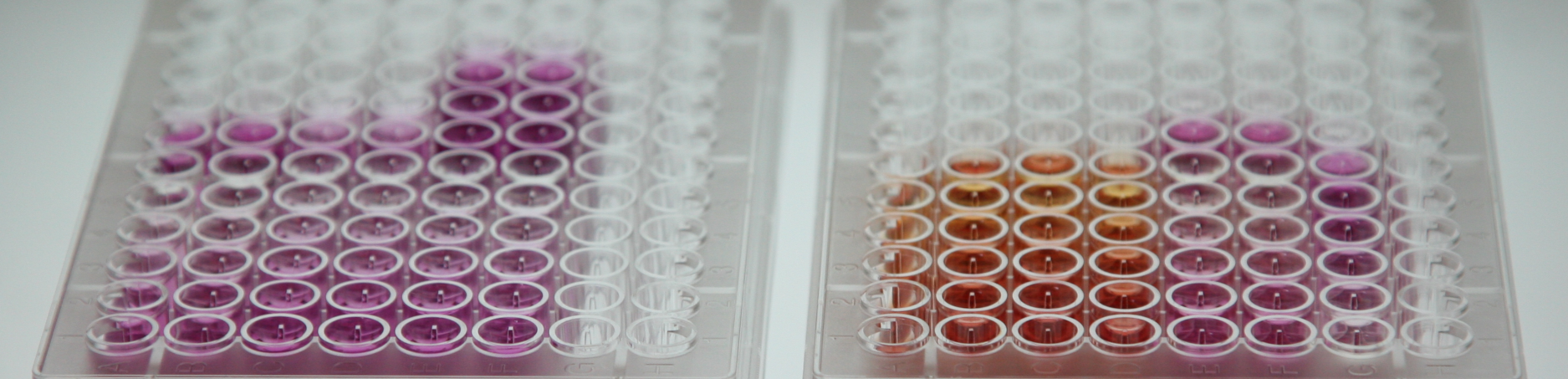
The purpose of this grant is to provide a functional and molecular definition of G0-like states in GBM tumors and their responses to SOC. In Aim 1, we will define molecular networks governing long- and short-term quiescent states in GBM patient tumors using a novel G0 reporter system in combination with single cell genomic analysis. In Aim 2, we will test the hypothesis that dormant G0 GBM cells have unique RNA and chromatin signatures required for SOC survival and tumor regrowth. In Aim 3, we will study and nominate the NuA4/KAT5 lysine acetyltransferase complex as a key regulator of G0-like states in GBM and candidate therapeutic target. The Aims are built on strong preliminary data, including: the creation of a machine learning-based method for identifying G0-like cells in gliomas, integrated analysis of single cell RNA and chromatin analysis of primary and PDX GBM tumors with standard of care, a functional genomic screen to identify regulators of GBM G0, and key experimental models to functionally dissect G0 states in GBM tumor models. If successful, this grant will produce a new working model for GBM G0-like states, provide key genes and gene networks associated with G0, and analysis tools for identifying G0-like states in clinical samples. It will also define how these populations respond to SOC and shift tumor dynamics during recurrence. Finally, it will provide data for a new therapeutic strategy, "downgrading", where grade IV tumors are made less aggressive by triggering extended or permanent G0-like states in tumor cells.
Richard G. Ellenbogen, MD, FACS (Co-Investigator)
Sponsor: NIH/NINDS